


GenScript cPass™ SARS-CoV-2 Neutralization Antibody Detection Kit (FDA EUA)
GenScript cPass™ SARS-CoV-2 Neutralization Antibody Detection Kit, is faster, easier, more scalable and automatable alternative to the traditional neutralizing antibody tests, such as virus neutralization test (VNT), pseudo-virus neutralization test (pVNT) and plaque reduction neutralization test (PRNT).
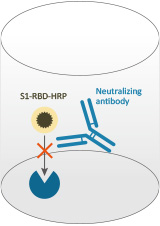 |
A new coronavirus, SARS-CoV-2, has recently emerged to cause a human pandemic. There is an urgent need for a robust serological test to detect neutralizing antibodies to SARS-CoV-2.There are two types of antibody tests that aim for detecting COVID-19 infection with sufficient specificity and sensitivity. The first type is the virus neutralization test (VNT) which detects neutralizing antibodies (NAbs) in a patient's blood. VNT requires handling live SARS-CoV-2 in a specialized biosafety level 3 (BSL3) containment facility which is tedious and time consuming, taking 2-4 days to complete. Pseudovirus-based virus neutralization test (pVNT) is similar, but still requires the use of live viruses and cells although handled in a BSL2 laboratory.The second assay type includes ELISA and lateral flow (rapid tests) for antibody detection. However, most of these assays only detect binding antibodies that have been generated to various epitopes of the whole virus and do not differentiate for the virus blocking Nabs.GenScript cPass™ SARS-CoV-2 Neutralization Antibody Detect\ion Kit is based on the Surrogate virus neutralization test (sVNT) established in Professor Lin-Fa Wang's lab at Duke-NUS Medical School. This test detects NAbs, but without the need to use any live virus or cells and can be completed in 1-2 hours in a BSL2 lab. Using purified receptor binding domain (RBD) protein from the viral spike (S) protein and the host cell receptor ACE2, the test is designed to mimic the virus-host interaction by direct protein-protein interaction in a test tube or an ELISA plate well. This highly specific interaction can then be neutralized, i.e., blocked by highly specific NAbs in patient or animal sera in the same manner as in a conventional VNT. |
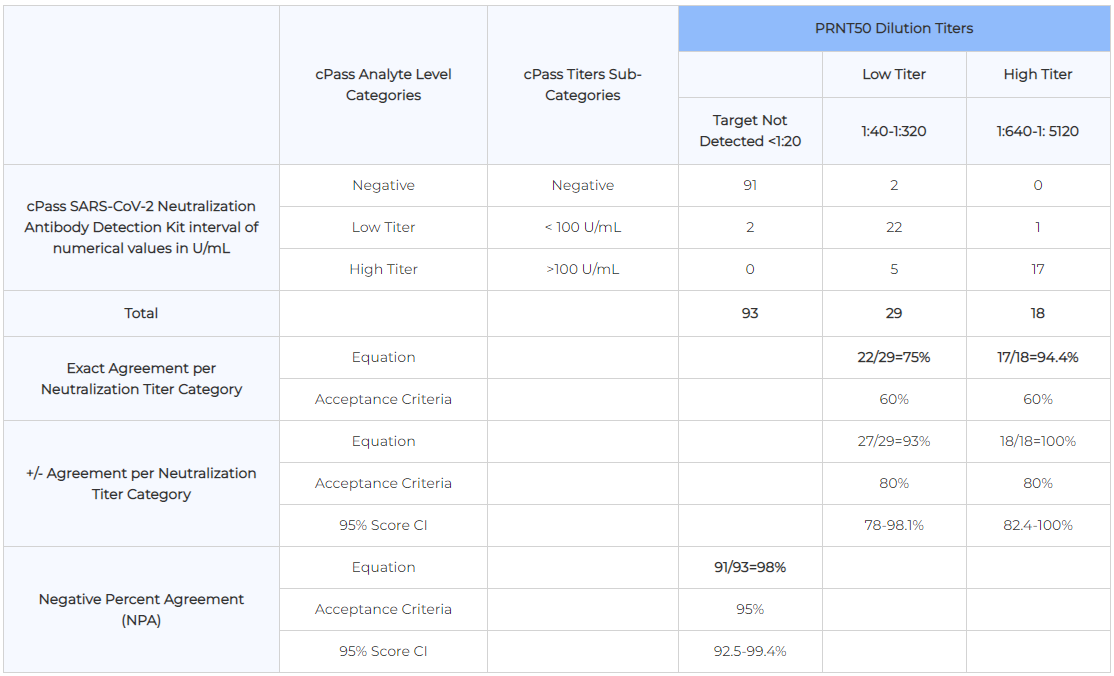
| PRNT50 Result Interpretation | |
| Positive: > 1:20 | Negative: < 1:20 |
| PRNT90 Result Interpretation | |
| Positive: > 1:10 | Negative: < 1:10 |
| PRNT50 Clinical Agreement | |
| PPA: 100% | NPA: 100% |
| PRNT90 Clinical Agreement | |
| PPA: 100% | NPA: 100% |
| PRNT50 Clinical Agreement | |
| PPA: 95.7% | NPA: 97.8% |
Lot # |
Original Expiration |
Extended ExpirationNew |
|---|---|---|
A210506 |
November 16, 2022 |
February 16, 2022 |
A210603 |
December 2, 2022 |
March 2, 2022 |
A210604 |
December 6, 2022 |
March 6, 2022 |