

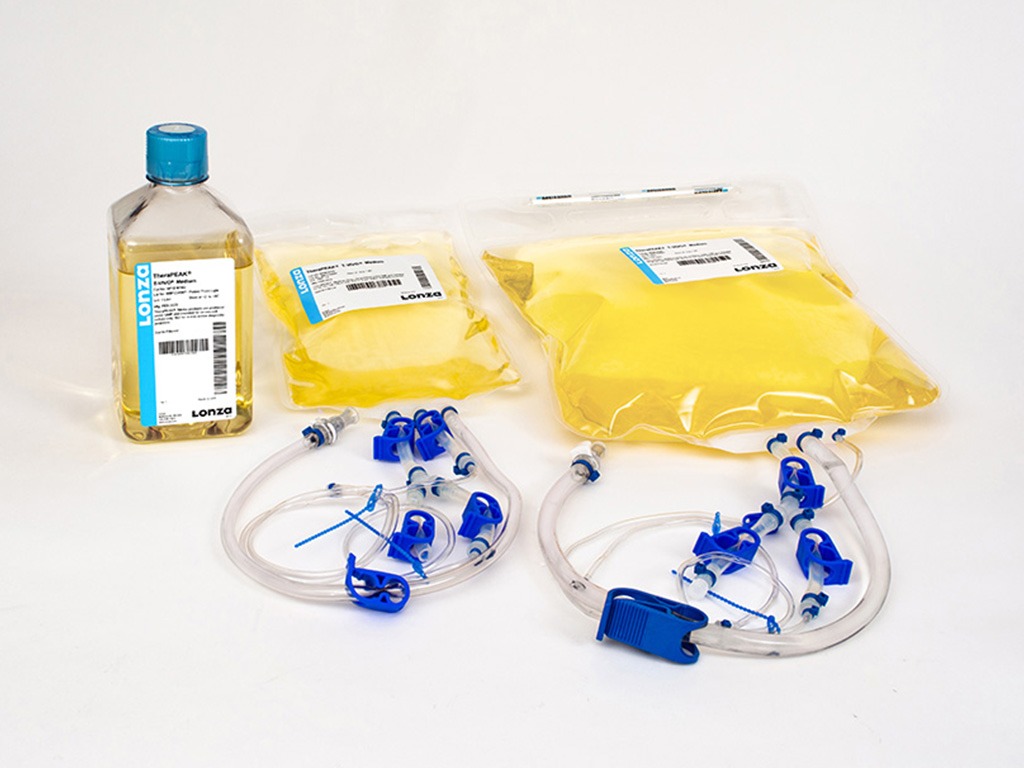

Lonza TheraPEAK® T-VIVO® Cell Culture Medium (Cat# BP12-970Q)
TheraPEAK® T-VIVO® Cell Culture Medium, chemically defined, non-animal origin (NAO), serum-free, with recombinant proteins, 1 L bottle
TheraPEAK® T-VIVO® Cell Culture Medium reduces variability concerns and enhances T-cell culture processes by enabling greater cell proliferation and viability, leading to increased process efficiency.
Its novel chemically defined formulation is serum-free and non-animal origin (NAO) to ensure consistency and provide increased process control.
With traceability documentation available including Certificates of Analysis and a Drug Master File, TheraPEAK® T-VIVO® Cell Culture Medium simplifies processes, supports regulatory compliance, and scale up from preclinical development through to manufacturing.
TheraPEAK® T-VIVO® Cell Culture Medium is available in the following sizes:
Customization of packaging for specific applications is available upon request. Please contact us for additional information.
All TheraPEAK® Products are produced according to applicable GMP standards and follow the USP/EP guidance for cell and gene therapy raw materials. TheraPEAK® Media Products are produced at FDA registered manufacturing sites under the auspices of ISO 13485 certified Quality Management Systems.
With demonstrated performance across a range of platforms including static T-flasks, spinner flasks, gas-permeable cell expansion devices, rocking platform bioreactors, stirred-tank bioreactors and the Cocoon® Platform, TheraPEAK® T-VIVO® Medium allows you to successfully generate patient treatments for numerous indications.
TheraPEAK® T-VIVO® Medium enhances the culture and expansion of:
Please email us at cs@medikonia.com for any enquiry. To place an order, please include the catalog number(s) of the product(s) in the email.